
Data Acquisition Critical to Imaging System that Monitors Brain Activity

NeuroPDA imaging system
A high-speed data acquisition system plays a critical role in an imaging system being used by researchers to monitor brain activity. The basic idea is to apply to brain tissue a dye (e.g. voltage-sensitive dye or Calcium dye) that lights up when neuronal activity occurs or simply use the intrinsic light change of the tissue during such activity. This method provides the ability to monitor large areas of brain activity at a much faster rate than possible with other monitoring methods such as magnetic resonance imaging. The device also monitors electrical signals from individual neurons. So researchers can, for example, provide an odorant stimulus to a mouse and determine exactly which areas of the brain respond. The NeuroCCD and NeuroPDA imaging systems produced by RedShirtImaging of Decatur, Georgia, are used by hundreds of neurobiologists, physiologists and biophysicists. The instruments also have been used clinically on an experimental basis and this application may someday dramatically improve the effectiveness of brain surgery. A critical factor in the design of these instruments was the need to acquire imaging data at the very high rates required to capture fast brain activity. A data acquisition processor (DAP) with onboard intelligence from Microstar Laboratories collects data from a photodiode array frame with 472 channels at a sample rate of up to 1.7 Kfps, making it possible to capture all the data that the camera can generate.
Acquiring Imaging Data at Very High Rates
A critical factor in the design of these instruments was the need to acquire imaging data at the very high rates required to capture fast brain activity. A DAP with onboard intelligence ...mak[es] it possible to capture all the data that the camera can generate. [And] onboard intelligence eliminates the danger of losing data regardless of the number of computer cycles dedicated to the foreground application or even if Windows crashes.
The human brain weighs only three pounds but is estimated to have more than 100 billion cells, mostly neurons. A neuron is basically an on/off device that is either in a resting state or is shooting an electrical impulse and transmitting it via synaptic connections to its neighbor, probably another neuron. Neurons connect to each other and form complex neural networks, which transmit information to other neurons or muscles. The signals that neurons transmit to each other are binary in nature, much like the bit stream with which computers communicate – or a complex Morse code. A nervous impulse involves the transmission of an electrical change along the membrane of a neuron. This nervous impulse or action potential, is a propagated, sudden and rapid change in transmembrane potential. Normally, the neuron membrane is polarized at rest, with a negative potential (-70 mV). The action potential consists of a rapid reduction of the negativity of the membrane to 0 mV and the inversion of this potential to values up to some +30 mV, followed by a very quick return to a value more negative than the potential at rest of -70 mV. These events occur in less than 5 milliseconds.
New ways to measure brain activity
A decade or two ago, the only way to study brain activity was to connect a probe directly to an individual neuron and measure its electrical activity. The problem with this approach was that with an individual probe required for each neuron measured, only a very small number of neurons were measured in a single experiment. Often, researchers want to determine which neurons are emitting interesting signals. To do this, the activity of many neurons must be measured simultaneously. Over the last decade, methods have been developed in which dyes are injected into the brain tissue; these dyes respond to electrical activity by either changing to a different color or emitting a fluorescent light. Then, a high-speed imaging system records the activity throughout the region of the brain exposed to the dye. At the same time, electrodes may be attached to individual neurons in order to obtain additional data.
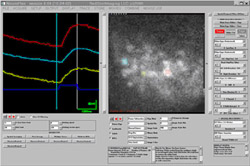
NeuroPlex imaging software
One of the greatest challenges faced by early researchers in this field was acquiring the huge amount of data produced by photodiode arrays that record brain images. A typical camera might record an 80 by 80 pixel frame at 16,384 intensity levels at a rate of up to 2,000 Hz, generating over 400 MB of data per second. Due to low cost and familiarity, the Windows operating system runs on the hosts of most imaging systems, and its limitations add to researchers' challenges. Windows is not a real-time operating system, making the imaging system vulnerable when Windows becomes occupied with other tasks, possibly interrupting the data flow. Windows uses up many cycles on the PC platform and when it takes control of the CPU it holds onto it for a long time in data acquisition terms. If the data acquisition process depends on the host it is easy to lose data while the operating system responds to the needs of the foreground application as it records data to disk and presents online results and analysis.
Developing tools to meet the challenge
Researchers attempting to use these early methods were forced to build their own equipment, a time-consuming and expensive process. In many cases, they could not build data acquisition systems capable of acquiring all the data generated by the latest photodiode and charge coupled device (CCD) cameras. RedShirtImaging was created to develop state of the art, high-speed, high-sensitivity imaging systems for the biological community. A group of leading scientists in the field founded the company to provide a combination of hardware and software in fully integrated packages. In developing their camera, data acquisition was one of their most critical design issues.
After investigating different data acquisition technologies, said Chun Falk, General Manager of the company, the designers discovered the technology of onboard intelligence. This extends the power of the Windows user interface by executing all processor-intensive routines in real time on the data acquisition board so that software on the PC can handle more demanding applications than usual. Onboard intelligence eliminates the danger of losing data regardless of the number of computer cycles dedicated to the foreground application or even if Windows crashes. The onboard processor continues to run the special routine that collects data from the acquisition devices totally independent of the PC. The designers selected a data acquisition processor (DAP) board with onboard intelligence from Microstar Laboratories, Bellevue, Washington. The DAP 4200a board acquires 472 channels of data from the photodiode array at up to 1.7 kiloframes per second. The DAP 840 with 8 channels is used to collect data in synchrony with the CCD camera, which has its own A/D card and frame grabber. Digital output channels on both cards open the camera shutter and provide the stimulus to the brain cells.
New products simplify researchers' task
RedShirtImaging has developed two main products. The NeuroPDA II uses the WuTech H469IV photodiode array system that provides a speed of 1.6 KHz and a 24 x 24 pixel frame and dynamic range of greater than 17 bits. The photodiode array system is designed for high-speed data acquisition at 0.9 millisecond per frame, which includes 464 diodes and 8 electrodes. This unit is designed for use with experiments that generate relatively high light levels because photodiodes do not saturate at light intensities achieved in neurobiological experiments. The system integrates all of the necessary components including the photodiode array, amplifiers, multiplexers, analog to digital converter, host computer and output panel. The system can also control a shutter and stimulator. The NeuroCCD imaging system is, on the other hand, designed for applications with lower light levels that do not saturate a CCD camera. The SciMeasure Analytical Systems camera has an 80 x 80 pixel frame with a full-frame rate of 2,000 Hz and 3 x 3 binning frame rate of 5,000 Hz.
Scientists at many leading research institutions already use these instruments. For example, Matt Wachowiak and Larry Cohen of Yale University collected intrinsic imaging signals with a NeuroCCD in response to four-second odorant pulses. Hiroyoshi Miyakawa, Masashi Inoue and Yoshihisa Kudo of Tokyo University of Pharmacy and Life Sciences injected the calcium indicator Rhod2 with a patch pipette and detected Ca transients induced by sodium action potentials using a frame rate of 2.0 KHz on a NeuroCCD. NeuroPDA photodiode imaging systems have also been used in a wide range of applications, such as pseudocolor display of a control response and epileptic activity in brain slices. In this experiment by Rezan Demir and Meyer Jackson of the University of Wisconsin, the epileptogenic slice showed a large secondary response that was not present in the control slices. In an example of the nonneural applications of the NeuroPDA, Ana Lia Obaid, Tetsuro Sakai and Brian Salzberg at the University of Pennsylvania used the instrument to measure the spontaneous activity of ganglions in the submucous plexus in a guinea pig small intestine.
Browse other customer applications.